|
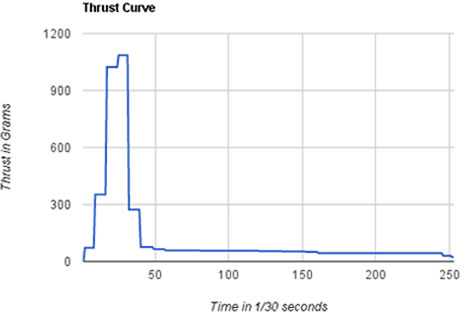
We tested our rocket engine!! There was one really big problem(which was fixed), but it went mostly well. The big problem was that our nozzle throat was 3/16" which was causing all of the other motors to CATO(catastrophe on takeoff).Thankfully, Andrew fixed this problem for us by enlarging it to 7/32", which worked well. As you can see, I have a video of the test that we did, and the device that measured our thrust. From that video, we made this graph, which is a thrust curve in grams for our engine. In the curve, It shows that our rocket had a lot of thrust in the beginning, but lost thrust and began to put out just enough to coast. I think that this would make a good motor for a small rocket, especially if you wanted a long flight time. The burn time for this motor is 8.43 seconds, and the total thrust(in grams) is 32196. We got all of this info from the thrust curve, as I said earlier. Personally, I wanted to use our motor and build a very small rocket, but my group decided that they wanted something different for the rocket, so we were not going to use this one. However,the engine we were going to use had a 3/16" nozzle size, which we determined to be to unstable. Now, we're planning on using the engine that we made.What they decided on was a rocket that would give us a lot of air time, and it looks good for that purpose. This engine and rocket combo could get really high, and I'm excited to build and launch the final rocket.
To catch up on some points, ( and also read something cool) I decided to do a science article review. I found an interesting article from he science board on reddit:
http://noosphereventures.com/new-substance-may-help-us-breathe-underwater-and-on-mars/ I don't quite know what subcategory this would be(perhaps biology), but it looks really cool. Apparently, researchers at the University of Southern Denmark created a cobalt based material that, quote, "not only draw oxygen directly out of the surrounding air of water, it can also release the stored oxygen back into it’s environment in a controlled manner when triggered to do so." There are many potential applications for this, including the use of this substance to make more oxygen on Mars, making new masks for deep sea divers, and helping people who have emphysema live more comfortably. THIS IS FREAKING EPIC!!!! This stuff could revolutionize the way we plan to go to the moon or Mars, and it could make it so we could finally explore all of the ocean floor. There's so much that could be done with this, its amazing!! Granted, I have not read the Abstract for the paper, so it may not be as wonderful as all that, but that's still pretty cool. We may not be able to go to mars or other planets yet, but I think this is a big step. Today we learned how to create water out of hydrogen. We did this so we could understand the reactions between the elements, and their various structures. I personally wanted to do this so we could design our rocket engine better, and so I could get some much needed points. Thankfully, the experiment went without a hitch.
The first thing we had to do was create a way to capture hydrogen. We did this by creating a citric acid solution and putting small magnesium pieces in the solution. After we poured the magnesium in, we had to very quickly put a balloon over the top of our glass. The magnesium caused the hydrogen in he solution to bubble up and get caught in our balloon, which began to fill up. After the solution stopped bubbling, we tied off the balloon, and put it up to a candle. The balloon(as it was filled with hydrogen) exploded, and when we looked at the inside pieces, we saw water droplets. I think this happened because of the fact that the balloon exploded. Since the balloon was filled with hydrogen, it reacted to the flame explosively, and when it exploded , the explosion released all of the hydrogen in it, along with a lot of excess energy, which made the hydrogen combine with the oxygen molecules in the air to create H20(water). I think that we got a really good experience out of this for multiple reasons. One, we get to understand reactions between atoms better, so we can design better rocket engines. Two, a movie called The Martian came out recently, and this process was similar to the way he made water in the movie, so we can fact check the science in that movie, and finally, if we ever need to make water, we have a way to now. I was in a loose partnership with Nancy and Fehung, and we worked pretty well together. As usual, no real problems with either of them. If we were going to do this again, I would want to try a bigger balloon and more magnesium, so that we could see approximately how much water it makes with certain amounts of magnesium. All in all, this was a fun and informative experience. As I started Section 3, I made a plan for what I was going to do with my time(learned my lesson from last section). As you can see above, in these next 2 weeks I'm hoping to do part 1 of the the rock project, some worksheets, and do one of the fuel activity with Andrew. 2 weeks from now, I'm going to post another one of these, and report on where I'm at overall.
This week, I started working on Stage 2 of the rocket project, which is designing engines for our rocket in the final stage. My partners are Fehung and Nancy, who are going to be my partners from now on. I'm excited to work with them, and building the rocket should be fun. In terms of what's happening now, we've started to research on how to make an efficient rocket engine for our future rocket. We did quite a bit of research on how to make it, and I learned a lot of stuff. For one, I learned that Black powder burns faster and has more thrust, while sugar rockets have the potential to be better, but they're hard to perfect. I also learned that while core burners usually have better acceleration, end burners have a longer burn time. We did a lot more research, but this is the most important information. Based on that research, we've determined that we want to do with the motor. We want a 25g black powder motor with a half core. The weird part about it is that we want it to be cored a quarter 3/16 and a quarter 1/16. We think this combination will give us a good thrust curve, but we're going to test for sure next week.
UPDATE:: We kicked the week off with this experiment, called "Out of Space & Time". The goal of this project is to look at something that is beyond our personal perceptions, whether it's beyond our scale of time, or out of scale of space. My partner( Fehung ) and I decided to do Out of Space. The pictures you see above are pictures of rust particles that were in the fume hood. We chose these spontaneously, but they were really cool to look at.
To take these pictures, we used the macro lens that Andrew provided and Fehung's iPhone. We took the container of of rust and poured some of it onto a blank white paper. To me, at least, it looks a lot like coffee beans, except with a more orange color. This was interesting to me because we always see rust on the side of buildings, on our bikes, and on our cars. In other words, we always see it as a part of something. It's weird to see it just on its own. While we were taking the pictures, I wondered whether or not rust forms differently on different substances. For examples, would rusted iron look different from rusted copper? Rust is caused by the atoms in the metal bonding with the oxygen in the air(or water). I think that the oxygen might bond differently if the metal was a different substance. Thus, if I were to investigate more, I would get specific substances and take pictures of them in different stages of rusting. My main takeaway from this was the realization that things always look different under a lens, not just bigger. It was a real eye-opening experience for me. |
AuthorAlex (Zachary)Wessel Archives
October 2017
Categories |


 RSS Feed
RSS Feed
